Patents incentivise research by giving entrepreneurs a temporary monopoly of the new knowledge they generate. But, in the pharma industry, it can take more than a decade from filing a patent to getting approval for a drug to be used in practice. That drastically shortens the amount of time a firm can use its monopoly. Supplementary Protection Certificates (SPCs) were developed as a simple means to extend the patent life of medicinal products. But it turns out that the application of the SPC regulation is not simple, and there is a lot at stake in terms of innovation and health care costs.
Big Pharma has a problem recouping R&D costs
It can cost as much as US$ 2.6bn for a pharmaceutical company to develop and prepare a drug for regulatory approval. It has not only to demonstrate its effectiveness but also its safety.1 However, fewer than one in twenty such drugs make it to market.2 Only half of the possible patent life (or even less) may then remain to recoup the R&D costs for those drugs that survive the clinical trials.
When pharma patents terminate and supplementary protection mechanisms kick-in heated policy debates are certain
This was less of an issue in the past when big pharma firms could rely on their ‘blockbuster’ drugs – that is, drugs addressing common diseases and selling over $1bn per year – for much of their sales. But the blockbuster drug patents have been expiring and the pharma companies are struggling to replace them with new, high-revenue products. Faced with this ‘patent cliff’, they have started to look for new ways to recoup R&D costs, including the management of patent life.
SPCs seem to offer a solution
The EU’s SPC regulation was intended as a simple response to this problem (and also as a means to foster European industry). It offers a seemingly straightforward way to extend patent life by up to five years. This may not seem long, but as SPCs usually kick in when a drug is in its most profitable phase – after the initial marketing efforts have started to pay off – even short patent life extensions, even months, are valuable.
The complexity of the SPC system arises from several factors, such as:
- An SPC does not simply prolong the life of a patent. Instead, it prolongs the patent protection for a medicine – that incorporates a patented substance. This subtle difference gives rise to a number of issues.3
- Yet another issue is the question of whether SPCs can be applied to second medical use or ‘indication’ patents.4 In the seminal Neurim case, the CJEU affirmed that it is possible to take out an SPC also on these.
But they could lead to unwanted effects
Opponents of the SPC system fear that because of this and other CJEU rulings, companies will, as it is said, “abuse” the system by taking out multiple SPCs on what is essentially the same product, prolonging patent protection well beyond the five years defined in the SPC regulation. The result would be less innovation and higher health-care costs.
The situation could be further aggravated by other regulations that allow time-limited monopolies. On top of an SPC, a pharma firm can also obtain a paediatric extension for up to six months, if a paediatric investigation plan is approved. This is intended to encourage research into the use of already-developed drugs for use with children, an otherwise smaller and less developed market. And for rare (so-called ‘orphan’) diseases affecting fewer than one in 20,000 people, it is possible to obtain up to ten to 12 years of market exclusivity for each orphan indication. There is also eight years of data exclusivity for the originally approved medicinal ‘reference’ product, during which time, generics firms cannot refer to clinical trial data to obtain marketing approval, plus two to three years of market protection.
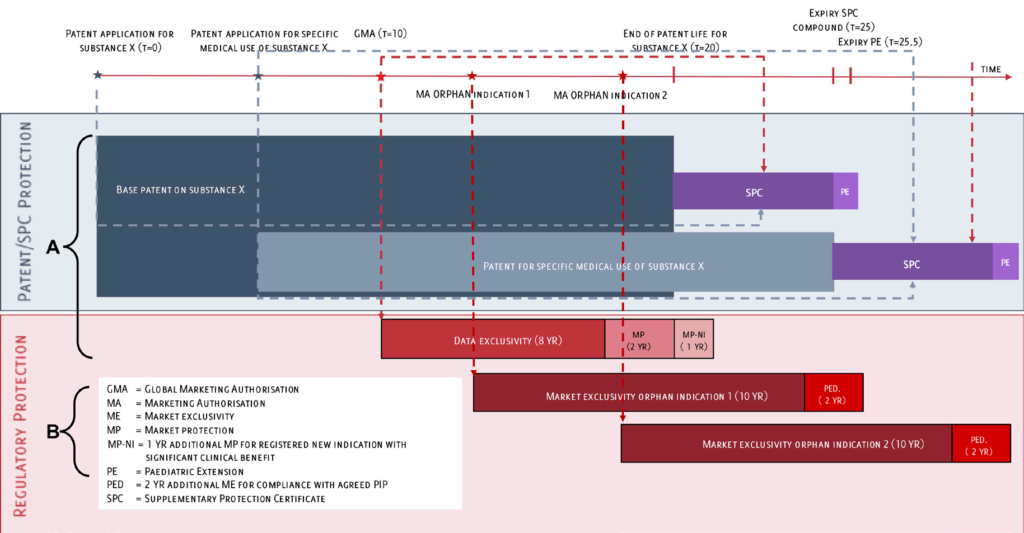
Blue represents patent protection; Red represents regulatory protection. The purple colour for the SPCs indicates the combination of features of patent and regulatory systems. Dotted lines show the relationships between patent application and marketing authorisation dates and how they relate to certain protection mechanisms. The width of the boxes (and the positioning of the two boxes exemplifying the patents) represents the scope of protection provided by the respective instruments
Does the scope of such additional protection need to be modified?
A recent report analysed the interaction of all these additional protections at European and national (Dutch) level.5 Whilst it did find that it is possible to stack up to different protection mechanisms (see figure above), it did not find evidence of widespread outright abuse of the system. Within the limits of the law, however, boundaries have been tested. It is clear that both intended and unintended effects can arise from the supplementary instruments. Certainly, the broadening of the interpretation of the SPC regulation through CJEU decisions seems to go well beyond the lawmakers’ original intentions. It means that SPC applications have to be more closely scrutinised than before and exacerbates the shortage of experts in the field who combine the needed complex regulatory and patent know-how.
It seems that the SPC regulation has fallen short of its target to foster the European pharmaceutical industry, not the least as U.S. and European firms alike can take advantage of the SPC system. In May 2018, the European Commission proposed an amended SPC regulation, introducing an export waiver. This controversial measure would make it possible for European generics companies to manufacture products for export purposes to third countries, which are otherwise protected by SPCs in the EU and hence could strengthen European generics firms. The story continues.
1 – Joseph A. DiMasi, Henry G. Grabowski, Ronald W. Hansen, Innovation in the pharmaceutical industry: New estimates of R&D costs, Journal of Health Economics, Volume 47, 2016, Pages 20-33.
2 – The probability of technical and regulatory success (PTRS) is estimated at 4.1% (see Paul SM, et al. How to improve R&D productivity: the pharmaceutical industry’s grand challenge. Nat Rev Drug Discov. 2010;9:203–214.)
3 – For example, medicinal products can incorporate several patents into one product. The question then is whether, for the same medicinal product, several SPCs can be obtained, each invoking a different patented aspect (the answer, sustained by the ECJ, is yes).
4 – Patent law allows not only for a particular substance with medical effect to be patented – it allows also for subsequent patenting of specific newly discovered indications (uses) of that same substance; or specific production processes for that substance; or for different formulations (e.g., pills vs. injections).



